Our research. |
Why study weird sex?
Mendelian inheritance is the standard inheritance model in evolutionary biology, even though there is growing evidence of frequent violations of it in nature. These distortions of segregation play out on various levels, fueled by genomic conflicts. For example selfish genes and chromosomes can boost their transmission probability via meiotic drive, or whole paternal haplotypes can be eliminated in paternal genome elimination. Even more familiar systems such as asexuality or Y chromosome inheritance are in fact extreme cases of segregation distortion; in other words, non-mendelian inheritance effects to certain extent probably all genetics systems in nature. In spite of this ubiquity, the evolutionary and genomic consequences are poorly understood and mostly focused on individual cases. In our laboratory, we research non-Mendelian inheritance in species with unusual reproduction modes using genetic, cytogenetic, epigenetic, and genomic approaches.
Unconventional chromosome inheritance
Genome elimination
The laws of genetic inheritance tend to be fair and symmetric in sexual reproduction: a son or a daughter receives one copy of their chromosomes from each of their parents and are equally likely to pass them on independently of the parent they got them from. Although this rule of fair chromosome inheritance is widespread, our group is interested in the weird and bizarre exceptions that have arisen in the tree of life. One of such exceptions is Paternal Genome Elimination, where males start their lives as diploids but eventually lose their paternally-inherited chromosomes either early in development or during spermatogenesis, so that they only transmit the chromosomes they received from their mothers. Another exception are supernumerary B chromosomes, which cheat during meiosis to enhance their transmission rate over the fair 50% under Mendelian inheritance. The Ross lab employs different taxa where PGE has evolved independently--mealybugs, fungus gnats and the human louse Pediculus humanus--as model systems to try to understand the evolutionary and mechanistic aspects of non-canonical chromosome inheritance. We are also interested in the B chromosomes of Pseudococcus viburni, a PGE mealybug where paternally-inherited B chromosomes change sides during spermatogenesis and are transmitted with the maternal set to the next generation. We also develop theoretical models to understand the evolutionary dynamics of different modes of chromosomal inheritance across the Tree of Life.
The laws of genetic inheritance tend to be fair and symmetric in sexual reproduction: a son or a daughter receives one copy of their chromosomes from each of their parents and are equally likely to pass them on independently of the parent they got them from. Although this rule of fair chromosome inheritance is widespread, our group is interested in the weird and bizarre exceptions that have arisen in the tree of life. One of such exceptions is Paternal Genome Elimination, where males start their lives as diploids but eventually lose their paternally-inherited chromosomes either early in development or during spermatogenesis, so that they only transmit the chromosomes they received from their mothers. Another exception are supernumerary B chromosomes, which cheat during meiosis to enhance their transmission rate over the fair 50% under Mendelian inheritance. The Ross lab employs different taxa where PGE has evolved independently--mealybugs, fungus gnats and the human louse Pediculus humanus--as model systems to try to understand the evolutionary and mechanistic aspects of non-canonical chromosome inheritance. We are also interested in the B chromosomes of Pseudococcus viburni, a PGE mealybug where paternally-inherited B chromosomes change sides during spermatogenesis and are transmitted with the maternal set to the next generation. We also develop theoretical models to understand the evolutionary dynamics of different modes of chromosomal inheritance across the Tree of Life.
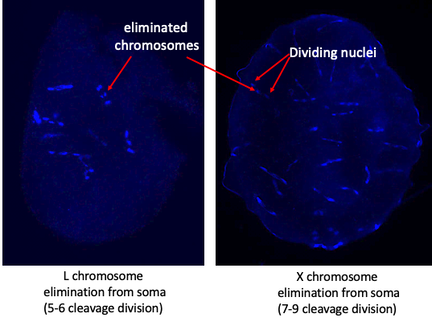
Germ line restricted chromosomes in fungus gnats
Chromosomes are usually present in all of the cells in animals, however there are exceptions to that. Fungus gnats have two large chromosomes that are found only in their germ line and get eliminated in all the somatic cell lineages - therefore called germ-line restricted chromosomes (GRCs). Previous cytological studies revealed that the two GRC chromosomes pair during meiosis and that they are rather big (~70Mbp each). In this project we investigate the origin and potential function of these chromosomes using genomic approaches.
Chromosomes are usually present in all of the cells in animals, however there are exceptions to that. Fungus gnats have two large chromosomes that are found only in their germ line and get eliminated in all the somatic cell lineages - therefore called germ-line restricted chromosomes (GRCs). Previous cytological studies revealed that the two GRC chromosomes pair during meiosis and that they are rather big (~70Mbp each). In this project we investigate the origin and potential function of these chromosomes using genomic approaches.
Alternative sex determining systems

Fungus gnats and maternal control over sex determination Fungus gnats are highly unusual in that females have control over sex determination. Mothers either produce only sons or only daughters throughout their life, and this difference is based on the presence of an inverted region on one of their X chromosomes. The molecular mechanisms behind this bizarre phenomenon and the evolutionary forces that have produced it are unknown. The atypical chromosome cycle these flies have also has interesting consequences for the evolution of sex chromosomes. We aim to understand how this strange system works, and what its evolutionary
causes and consequences might be.
Springtail double X system
The X chromosome evolution is rather special in species with Paternal Genome Elimination. There are only three known PGE clades to still have X chromosomes, although the sex determination was overwritten by other mechanisms - fungus gnats, gall midges and globular springtails. The X chromosomes are likely remains of the ancestral X chromosome from the old times of more conventional reproductive strategy. Interestingly enough, globular springtails don’t just have one X chromosome, they have two different X chromosomes (therefore could be called X1X200 system). Our recent preprint shows that the two X chromosomes represent a very large portion of the genome (https://www.biorxiv.org/content/10.1101/2020.10.04.325340v2), we further work to understand the peculiar evolution of the double-X system in globular springtail using comparative genomic and transcriptomic approaches.
Molecular mechanisms of paternal genome elimination in mealybugs
Some species have abandoned sex chromosomes entirely. Mealybugs that employ paternal genome elimination are either fully diploid females or fully haploid males, similar to ants, bees, and wasps (Hymenoptera). As with that system, sex of mealybug offspring is likely under maternal control; however, unlike Hymenoptera, haploid males reliable carry their maternally inherited alleles, with their father’s haplotype discarded. Our lab is working to identify the molecular mechanisms by which the paternal genome is identified and eliminated to create males.
Genomic imprinting and epigenetics
Genomic imprinting is the differential expression of alleles in diploid individuals, with expression being dependent on the sex of the parent from which the allele was inherited. Mealybugs display an unusual instance of whole genome imprinting called Paternal Genome Elimination (PGE), described [here](link to Unconventional chromosome inheritance). The mechanisms by which the paternal chromosomes in males are recognised, and thus condensed in the soma and eliminated from the sperm, remains unknown. Imprinted genes in mammals are mediated via epigenomic mechanisms such as DNA methylation and histone modifications. Mealybugs have particularly high levels of DNA methylation, for an insect species, along with sex-specific profiles of histone modifications known to be involved in silencing. Our lab is investigating the role of these epigenomic mechanisms, along with chromatin structure and ncRNAs, in the process of Paternal Genome Elimination.
In addition to an unusual inheritance system, mealybugs also show extreme sexual dimorphism and possess no sex chromosomes. It is therefore also predicted that epigenomic mechanisms mediate sex determination and sex differences. Our lab has recently demonstrated the mealybug, Planoccocus citri, shows an extreme sex-specific DNA methylation profile. We are now working to understand how chromosome specific epigenomic mechanisms change throughout development to begin to untangle the role of epigenomics in PGE, sex determination and sexual dimorphism.
In addition to an unusual inheritance system, mealybugs also show extreme sexual dimorphism and possess no sex chromosomes. It is therefore also predicted that epigenomic mechanisms mediate sex determination and sex differences. Our lab has recently demonstrated the mealybug, Planoccocus citri, shows an extreme sex-specific DNA methylation profile. We are now working to understand how chromosome specific epigenomic mechanisms change throughout development to begin to untangle the role of epigenomics in PGE, sex determination and sexual dimorphism.
The evolution of hermaphroditism
For sexually reproducing species, the evolution of separate male and female sexes carries some evolutionary downsides. Only one of the two sexes produces offspring, and then only after encountering and mating with a member of the opposite sex. In contrast, self-fertilizing hermaphrodites assure reproductive success on their own and even a single individual can found a new population. In spite of this apparent advantage, hermaphroditism is relatively rare in animals, and almost unknown in one of the most specious clades: the insects. Our lab studies one of the only three known hermaphroditic insects, the cottony cushion scale insect (Icerya purchasi). We seek to understand the system at multiple levels from how sperm and egg cells are made within the same individual to how hermaphroditism may have contributed to the the global spread of this species.





